Abstract
Background: Ormeloxifene is a non-steroidal, breast parenchymal selective anti-estrogen drug used for regression of fibroadenomas as well as breast nodularity and mastalgia. This study aims in redefining the role of Ormeloxifene in regression of fibroadenomas with respect to lump size volume and associated symptoms of mastalgia. Materials & Methods: It is prospective cohort study with a sample size of 98 patients of age less than 30 years, conducted at tertiary care center in Central India that includes clinically and cytologically diagnosed patients of fibroadenoma with size less than 5 cm. All enrolled patients were prescribed Tab. Ormeloxifene 30 mg alternate day for 3 months and the regression of lump size was evaluated by both clinically and ultrasonologically at the end of first, third and sixth month. Results: The mean age of participants was 20.71 ± 3.47years, with 61.4% patients presenting with single discrete lump. 38 patients (39.6%) patients were having the lump size between 1.6-2.0 cm3 at the time of presentation. At the end of 3 months, total regression of the lump was observed in 71 patients (73.95%). Complete regression was observed in patients having initial lump size of 2 cm3. None of the patient had any serious side effects of the drug, except for reversible oligomenorrhea or amenorrhea in 72% of patients, where the cycles became regular after completion of therapy. Conclusion: Ormeloxifene is a safe and effective drug that helps in regression of fibroadenomas, especially of size around 2 cm3, as an alternative to surgical excision.
Introduction
Fibroadenoma is most common benign tumor occurring in young females of age less than 30 years. It is also a cause of major concern and anxiety among unmarried females. Though causative factors of fibroadenoma are heterogeneous, estrogen has been linked as a major responsible factor. Surgical removal of fibroadenoma is described but it often leads to a bad and ugly scar, hurting patient both physically and psychologically. Ormeloxifene is a novel non-steroidal, selective anti-estrogen drug with an additive effect of being onco-protective to breast cells. According to previous studies [1,2], Ormeloxifene has shown promising results in regression of size of fibroadenoma, with almost 35% to 65% subjects benefitting with the use of Ormeloxifene. But at the same time, around 25% to 40% patients did not respond to tablet Ormeloxifene with no or minimal regression in size of fibroadenoma. The rationale behind the present study is to further define the role of Ormeloxifene in regression of fibroadenoma breast.
Materials and Methods
The study was a single-centered, prospective cohort study conducted at All India Institute of Medical Sciences, Nagpur after approval from Institutional Ethics Committee (IEC/Pharma/2021/279). The duration of study was from October 2021 to September 2023 with one year follow up. The sample size was 96, using OpenEpi system with sample size calculation based on considering the regression rate of fibroadenoma to be 65%, confidence level of 95%, absolute precision of 10% and 10% loss to follow up. All patients of age less than 30 years, with a clinically and cytologically diagnosis of fibroadenoma with size less than 5 cms, were included in the study. Patients with Polycystic Ovarian Disease (PCOD), patients who are on oral contraceptive pills, pregnant and lactating females and patients with breast lump post-surgical recurrence were excluded from the study. All patients underwent ‘Triple Assessment’ by clinical, cytological and ultrasound of breast. All patients satisfying the inclusion criteria, the lump size was calculated using vernier caliper in two greatest dimensions, right angle to each other and by ultrasonography. In patients with multiple lumps or bilateral lump, the lump with largest volume was considered as index lump. The volume of fibroadenoma in cubic centimeter will be calculated by using the simplified formula for an ellipsoid:
Volume of fibroadenoma= axbxcx0.52
Where, ‘a’ is the largest dimension, ‘b’is the dimension perpendicular to a and ‘c’ is average of a and b= a+b/2.
All the patients were prescribed Tablet Ormeloxifene 30 mg once daily for alternate day for three months after explaining in detail about the drug, its action and potential side effects. Patients were followed at the end of first, third and sixth month from the start of medication. Assessment of lump size was done at every visit using vernier caliper and ultrasound of the breast. Mastalgia, if present was recorded at the time of initiation of therapy using Visual Analogue Scale (VAS) and again at every monthly visit. Side effects of drug, like nausea, vomiting, allergic rash, presence of amenorrhea, oligomenorrhea or weight gain were also recorded on subsequent visits.
Regression Rate
Based on clinical examination of breast lump using vernier caliper supported by ultrasound of breast lump, regression in size of breast lump will be evaluated at 1 month, 2 months and 3 months. Complete regression/ more than 75% regression of lump at the end of 3 months will be considered as successful treatment. 25% regression in size of lump or less will be considered as failure of treatment and the patients will be offered the option for surgical excision of fibroadenoma. Patients with regression rate between 26% to 74% will be offered the option for either surgical excision or continuation of same therapy additional 3 months (Figure 1).
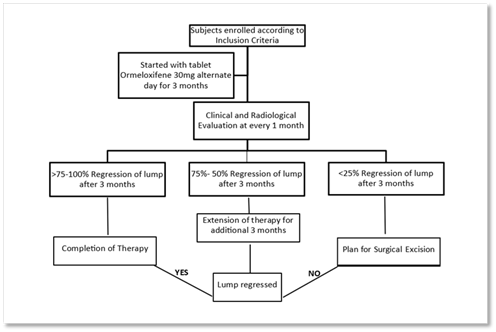
Statistical Analysis
Data were analyzed using SPSS version 24.0. Descriptive statistics such as mean & standard deviation were used for quantitative variables, and frequencies, percentages & Chi-square test were used for qualitative variables. ANOVA was used to compare the size and VAS scores over time intervals and Youden index was used with Receiver Operating Characteristic (ROC) curve to determine the optimal cut-off value and evaluation of performance of continuous variables.
Results
96 patients were enrolled in the study who met the inclusion criteria. The mean age of participants was 20.71 ± 3.47years. 59 patients (61.4%) presented with single discrete lump and in 37 patients (38.6%) multiple ipsilateral or bilateral lumps were present. 83 patients (86%) were nulliparous at time of enrollment. 38 patients (39.6%) patients were having the lump size between 1.6-2.0 cm3 at the time of presentation as shown in figure 2.
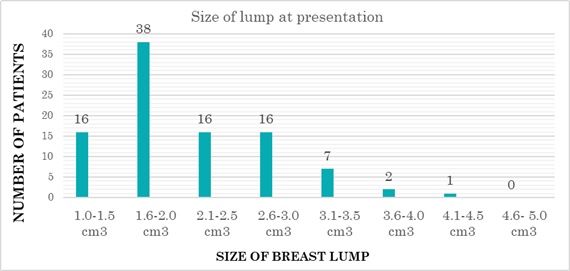
Only 20 patients (21%) presented with mastalgia, and out of these 20 patients 12 patients were having non-cyclical mastalgia.
After 3 months of therapy with Tab. Ormeloxifene 30 mg once daily for alternate days, total regression of the lump was observed in 71 patients (73.95%), whereas no regression was seen in 5 patients (5.20%). 20 patients (20.83%), out of 96 patients showed partial regression of fibroadenoma after completion of 3 months, and they were counselled for continuation of therapy for additional 3 months. Out of these 20 patients, only 6 patients showed complete regression after additional 3 months of therapy, total duration of therapy being 6 months, whereas rest 14 patients underwent surgical excision for fibroadenoma. It was noteworthy that the patients having fibroadenoma of size less than 3cms exhibit complete or partial response in terms of regression (Table 1).
| Size at Initial Presentation | No. of patients | >75 % to 100% reduction in size after 3 m | Percentage | >75 % to 100% reduction in size after 6 m | Percentage | Inadequate Response after 6 m |
| 1 – 1.5 cm3 | 16 | 16 | 100% | - | - | - |
| 1.6 - 2.0 cm3 | 38 | 38 | 100% | - | - | - |
| 2.1- 2.5 cm3 | 16 | 11 | 68.75% | +3 = 14 | 87.50% | 02 |
| 2.6 – 3.0 cm3 | 16 | 06 | 37.50% | +2 =8 | 50.00% | 08 |
| 3.1 - 3.5 cm3 | 07 | 00 | - | 01 | 14.28% | 06 |
| 3.6 - 4.0 cm3 | 02 | NIL | 0% | NIL | 0% | 02 |
| 4.1 – 4.5 cm3 | 01 | NIL | 0% | NIL | 0% | 01 |
| 4.6 – 5.0 cm3 | 00 | - | - | - | - | - |
| TOTAL | 96 | 71 | 73.95% | 06(77) | 80.20% | 19 |
Table 1 also shows that, there was no response to therapy in patients having fibroadenoma of size more than 3.5 cm3, whereas all fibroadenomas with size less than or equal to 2 cm3 showed 100% regression in lump size. When the maximum length and breadth of fibroadenoma was plotted by the Receiver Operating Characteristic (ROC) Curve, the analysis yielded an Area under Curve of 0.91(95% CI). The cut-off points for maximum diameter of the tumor size that shows complete regression was found to be 2.15 cm3 at which the Sensitivity was 100% and Specificity was 82.9% and Youden’s index (J) was 0.829 (SE:0.045) as shown in Figure 3.
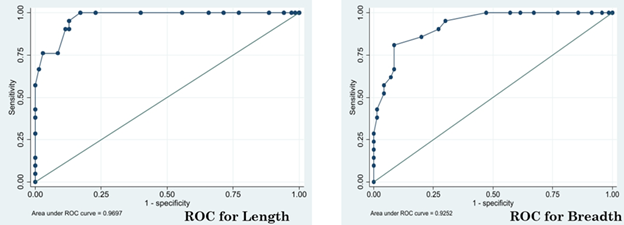
Mastalgia was found in 20 patients (21%), with a mean VAS of 4.45 ±1.02 at the time of presentation. Mastalgia was reduced in 16 out of these 20 patients (75%) after 3 months of therapy with mean VAS being 2.12 ±0.45.
Alteration of menstrual cycle with oligomenorrhoea or amenorrhea was the most common side effect of the drug affecting 69 patients (72%), followed by bloating of abdomen in 14 patients (14.5%) and dull-aching lower abdominal pain in 10 patients (10.41%). The patients with menstrual irregularities were followed up for 3 months after completion of therapy and all of them achieved regular menstrual cycles after stopping the drug. These patients were further followed up for one year and no recurrences were noted in patients who showed complete response to therapy after 3 and 6 months respectively.
Discussion
Fibroadenomas of the breast are discrete, solid, benign tumors considered under the spectrum of Aberrations of Normal Development & Involution (ANDI) of breast parenchyma [3] and occurs due to hyperplasia of Terminal Ductal Lobular Unit (TDLU). They are regarded as most common benign tumors occurring in breasts of females less than 30 years of age [4]. Most of the fibroadenomas are less than 3 cms in size, but occasionally they may grow beyond 5cms and then they are categorized as giant fibroadenomas [5]. Although, most of the fibroadenoma are painless, but some may be associated with cyclical or non-cyclical mastalgia and are perceived as mobile lump within the breast or breast mouse. Fibroadenomas can occur bilaterally or may present as multiple lumps in the same breast. They are more importantly cause of anxiety and apprehension among young females, who might fear of harboring breast malignancy. Spontaneous regression of these fibroadenomas in approximately 10-15 % of patients is well documented in literature [6]. Surgery should not be first option in management of small fibroadenoma as it may result in giving an ugly scar to a patient and in case of multiple fibroadenomas, multiple scar or loss of breast volume may give rise to asymmetry of breast leading to more anxiety, depression and psychosocial issues.
Cyclical changes in breast parenchyma are constantly influenced by phases of menstruation with enhanced dynamicity occurring during pregnancy and post-partum lactational period. Various hormone receptors for estrogen, progesterone and prolactin are demonstrated on breast tissue and this hormonal imbalance, especially excess of estrogen, leads to development of mastalgia and/or nodularity in young patients [7].
Mastalgia is a bothersome and challenging symptom to treat, as it gives rise to anxiety, psychosocial stress, interferes with sexual activity, imparts fear of malignancy and overall create a negative mindset for the sufferer [8]. It can be cyclical or non-cyclical mastalgia. Cyclical mastalgia is associated with estrogen surge during ovulation with elevation of pain in premenstrual period and is regressed after menses. Non-cyclical mastalgia is of less intensity and can be associated with musculo-skeletal abnormality or inflammation as seen in Tietze’s syndrome where location of pain is at inner quadrants of breast near costo-chondral joints, or maybe non-specific occurring near axillary tail of Spence [1]. Many pharmacological and non-pharmacological agents like tamoxifene, danazol, bromocriptine, oral and topical NSAIDs, Gamolenic acid, evening primrose oil have been tried previously for alleviation of mastalgia and breast nodularity with varying success and considerable side effects of steroid use like amenorrhea, weight gain, hirsutism and acne [8,9].
Ormeloxifene is a non-steroidal, Selective Estrogen Receptor Modulator (SERM) molecule also known as Centchroman was developed by Central Drug Research Institute, Lucknow, India and was included in National Family Welfare Program in 1995 as an oral contraceptive with added advantage of less frequent administration and better compliance among users [1]. It is commonly marketed in India by names of Saheli or Sevista or Novex [10]. Ormeloxifene has a strong anti-estrogen effect on proliferation of TDLU, but weak agonist effect on uterine endometrium [6]. It is remarkably devoid of steroid based androgenic effects like weight gain or hirsutism, does not alter the rate of fertility after stopping the drug and is secreted in scanty amount in milk, so making it safe for consumption even in lactating mothers with no deleterious effects on babies who are breastfed [9].
According to previous studies by Dhar et al [1], Brahmachari et al [8] and Rai et al [6], recommended dose for regression of fibroadenomas is Tablet Ormeloxifene 30mg alternate day therapy for 3-6 months, which is the same dose that was prescribed in our study. However, literature suggests that Tablet Ormeloxifene 30 mg twice a week prescription has quoted similar results in regression of breast lump and mastalgia as shown by Jeeva N et al [4] and Santosh CS et al [11].
In our study, the mean age of patients was 20.71 ± 3.47years, which suggest fibroadenomas occurs more commonly in younger age and 83 patients (86%) were found to be nulliparous, the results matching with the study conducted by Singla et al [12].
After 3 months of therapy with Tab. Ormeloxifene 30 mg for alternate days, total regression of the lump was observed in 71 patients (73.95%), whereas no regression was seen in 5 patients (5.20%). 20 patients (20.83%), out of 96 patients showed partial regression of fibroadenoma after completion of 3 months. Out of these 20 patients, only 6 patients showed complete regression after additional 3 months of therapy, making a total of 77 patients (80.2%) showing complete regression. These results were comparable with the study conducted by Jeeva et al [4]. The regression percentage was higher in our study as compared to previous studies by Jain et al in 2015 [13], who reported complete regression in 58.3 % patients, partial regression in 41.7%; Verma et al in 2016 [14], reported complete regression in 48.6% patients and partial regression in 51.4 % patients; Bhrahmchari et al in 2021 [8], reported complete response in 34% patients and partial response in 46.1% patients. The higher regression rate in our study is attributed to the higher number of included patients having lump size volume less than or equal to 3cm3. All patients with initial lump size on presentation less than or equal to 2 cm3 showed complete regression of lump with significant p-value of 0.001. Whereas, patient with lump size volume more than 3 cm3 showed no regression in lump size, even after 6 months of therapy. The patients with lump size volume between 2.1- 3.0 cm3 showed a variable response with regression of lump occurring in 87.5% patients with initial lump size volume between 2- 2.5cm3 whereas, only 50% patients showed regression after 6 months of therapy with initial lump size volume between 2.6-3.0 cm3. This indicates, smaller the fibroadenoma size, more are the chances of regression. The Receiver Operating Curve analysis for size of fibroadenoma, demonstrated that the chances of lump regression is higher if the cut-off margin is taken as 2.15cm3.
None of our patients developed any adverse drug reactions to tablet Ormeloxifene, although oligomenorrhoea or amenorrhea was seen in 69 patients (72%), which is quite higher as compared to Shukla et al, who reported the incidence of oligomenorrhoea to be 27.32% [9]. All our patients achieved normal menstrual cycles after completion of therapy on regular follow up visits.
The limitations of our study were a small sample size with short duration of follow up, as recurrence of fibroadenomas may take months to years and non-inclusion of subjects with non-discrete breast nodularity for Ormeloxifene trial.
Conclusion
Ormeloxifene, a novel non-steroidal preparation is safe and effective method for regression of fibroadenomas, especially in a young, nulliparous female with a breast lump size volume of around 2 cm3, with no major adverse drug reactions or side effects except for transient, reversible amenorrhea or oligomenorrhoea till the therapy is continued and is a very good alternative for surgical excision.
Declarations
Conflict of Interest
None
Funding Statment
None
Disclosure
The paper was presented at annual conference of Association of Breast Surgeons of India, ABSICON 2024 at Lucknow, India.
Acknowledgements
Not applicable.
References
- Dhar A, Srivastava A. Role of Centchroman in Regression of Mastalgia and Fibroadenoma. World J Surg. 2007; 31: 1178-84. DOI ↗ Google Scholar ↗
- Tejwani PL, Nerkar H, Dhar A, et al. Regression of Fibroadenomas with Centchroman: a Randomized Controlled Trial. Indian J Surg. 2015;77(Suppl 2):484-489. DOI ↗ Google Scholar ↗
- Dupont WD, Parl FF, Hartmann WH, et al. Breast cancer risk associated with proliferative breast disease and atypical hyperplasia. Cancer. 1993;71(4):1258-1265. DOI ↗ Google Scholar ↗
- Jeeva N, Radhi S, Ponvelavan R. To Study the Effect of Ormeloxifene in the regression of Mastalgia & Fibroadenoma. Int J Acad Med Pharm. 2023;5(4):623-7. DOI ↗ Google Scholar ↗
- Santen RJ, Mansel R. Benign breast disorders. N Engl J Med. 2005;353(3):275-285. DOI ↗ Google Scholar ↗
- Rai P, Nityanand S, Singh A, Singh SK, Singh N, Singh AK, Singh D. Role of centchroman in regression of fibroadenoma: A 2-arm randomized control trial. Clinics. 2025 Jan 1;80:100567. DOI ↗ Google Scholar ↗
- Hughes LE, Mansel RE, Webster DJ. Aberrations of normal development and involution (ANDI): A new perspective on pathogenesis and nomenclature of benign breast disorders. Lancet 1987;2:1316-9. DOI ↗ Google Scholar ↗
- Brahmachari S, Bhagat V, Patil P, Vasuniya V. Evaluating the Effect of Ormeloxifene on Multiple Fibroadenomas and Mastalgia. J Pharm Bioallied Sci. 2021 Nov;13(Suppl 2):1386-1389. DOI ↗ Google Scholar ↗
- Shukla V, Patel V, Gupta R, Kumar S, Gaharwar APS. Role of Centchroman in Mastalgia: An Experience in a Tertiary Care Hospital of Vindhya Region. IJSS Journal of Surgery 2017;3(3):22-26. DOI ↗ Google Scholar ↗
- Girish TU, Faraz M. Role of Ormeloxifene in regression of benign breast diseases. Int Surg J 2020;7:743-50. DOI ↗ Google Scholar ↗
- Santhosh CS, Mallikarjuna MN, Kumar HC, Naik S, Shamala S, Salman. To study the role of ormeloxifene in mastalgia and fibroadenoma of breast. Int J of Life Sciences, Biotechnology and Pharma Research 2023;12(3):1436-41. DOI ↗ Google Scholar ↗
- Singla NK, Bhatia R, Verma R, Bhatia SK. A prospective study on the role of centchroman in regression of Fibroadenoma. Clin Surg. 2021;6:3212. DOI ↗ Google Scholar ↗
- Jain, et al. Efficacy of ormeloxifene in the treatment of benign breast diseases: A randomized controlled trial. Journal of Mid-life Health. 2015;6(2):57-61. DOI ↗ Google Scholar ↗
- Verma, et al. Effectiveness of ormeloxifene in the treatment of breast fibroadenoma: A retrospective study. Indian Journal of Surgery. 2016;78(6):471-475. DOI ↗ Google Scholar ↗
